Hematopoietic Cell Therapy
Hematopoietic stem cells (HSCs) have long been used to provide life-saving treatments for patients with malignant and non-malignant hematological disorders. While these treatments originally took the form of conventional bone marrow transplants, more recent breakthroughs in the field have provided cell therapy researchers with methods to correct genetic mutations in a patient’s own cells for autologous HSC transplantation.
We have compiled a selection of scientific resources to help you on your way in the complex and exciting field of hematopoietic cell therapy.
CRISPR-Cas9 Editing of Hematopoietic Stem and Progenitor Cells
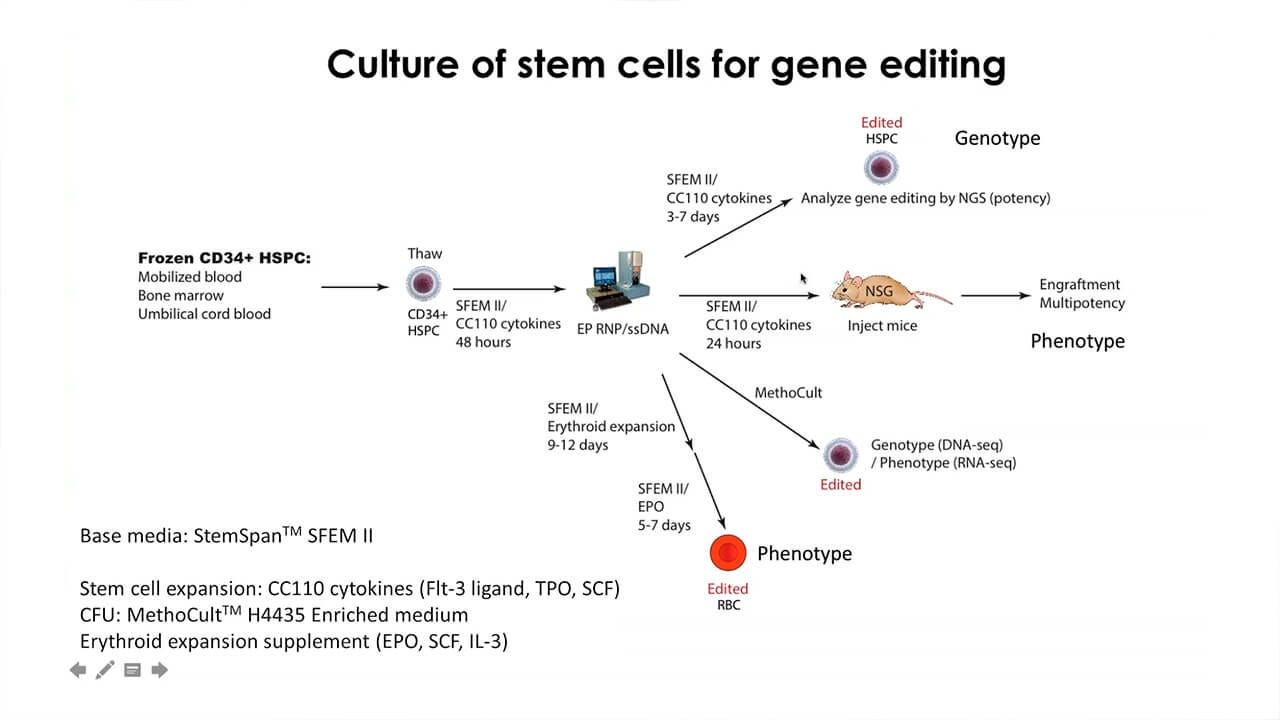
In this webinar, Dr. Mark DeWitt will discuss the use of gene editing techniques for hematopoietic stem and progenitor cells HSPCs. Topics he will include are:
- Delivery of gene editing machinery to target cells
- Homology-directed repair for precise editing
- Culture conditions required for efficient editing
- Levels of correction needed for the treatment of monogenic disorders, such as sickle cell disease
- Lost in Translation - Moving Your Research to Clinical TrialsThis webinar describes steps and considerations involved in translating research to the clinic.
- New Tools for the Ex Vivo Expansion of Human Hematopoietic Stem and Progenitor CellsThe development of standardized ex vivo HSPC expansion methods is needed to generate clinically relevant cell numbers for transplantation. This webinar will review key concepts in human HSPC biology, including standardized methods and recently demonstrated strategies for expanding HSPCs in culture. Additionally, ∫£Ω«∆∆Ω‚∞Ê products for the entire workflow of HSPC expansion will be covered, from isolation to expansion and differentiation, and analysis.
- How to Use Primary Cells to Develop the Next TherapyHuman primary cells are cells isolated directly from tissues, including blood and bone marrow. The use of human primary cells increases the physiological relevance of cell culture systems, enabling you to generate meaningful data that is more predictive of in vivo outcomes. Learn about the advantages of starting with human primary cells for your immunology and cell therapy research workflows. Key considerations for choosing the ideal starting material, donor characterization, regulatory challenges, and tips to streamline your cell-based assays with human primary and cultured cells are also covered.